Warning: Directory /usr/share/nginx/html/en/data/cache/URI not writable, please chmod to 777 in /usr/share/nginx/html/en/plugin/htmlpurifier/HTMLPurifier.standalone.php on line 15750
Warning: Directory /usr/share/nginx/html/en/data/cache/URI not writable, please chmod to 777 in /usr/share/nginx/html/en/plugin/htmlpurifier/HTMLPurifier.standalone.php on line 15750
Warning: Directory /usr/share/nginx/html/en/data/cache/URI not writable, please chmod to 777 in /usr/share/nginx/html/en/plugin/htmlpurifier/HTMLPurifier.standalone.php on line 15750
[Linked in] August 25th K-BPR : K-BPR Household chemical product subject to approval
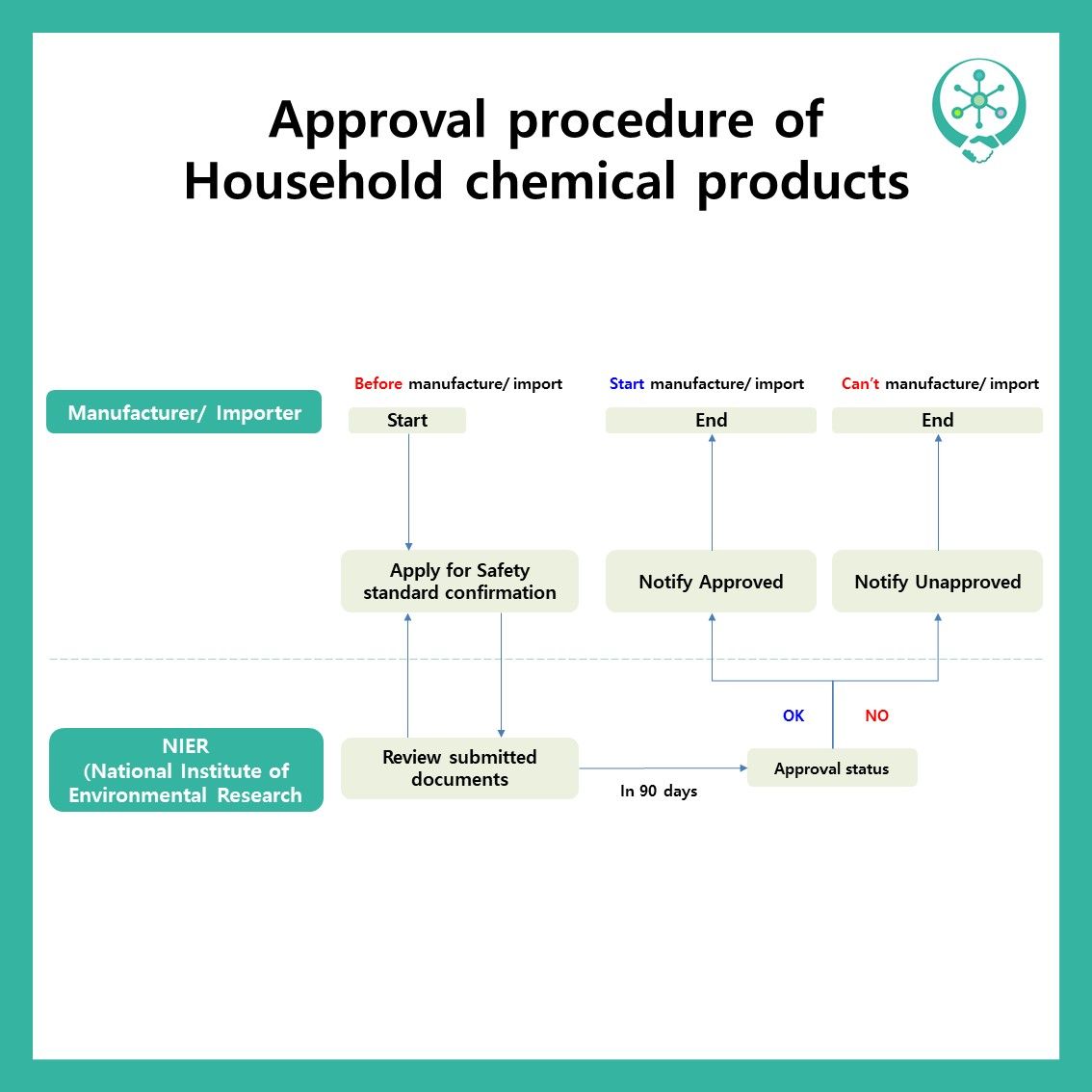
Good morning! We would like to look around the subject to ‘APPROVAL’ among the household chemical product subject to safety confirmation'. Under K-BPR article 10, the person who manufactures or imports the household chemical product subject to safety confirmation subject to approval should be verified by the Ministry of Environment. The items that should be approved are as below.
1) Sterilizing products: Antibacterial for humidifiers, disinfectants/sterilizers for preventing infectious diseases, disinfectants/other disinfectants for quarantine.
2) Relief products: Health relief, prevention, and inducement pesticides, health repellents/infectious disease prevention pesticides/infectious disease prevention fleshers
Check more details in the card attached.
Applications for approval of household chemicals subject to safety confirmation can be made to the National Institute of Environmental Research (http://chemp.me.go.kr), and submission documents include applications for approval, documents on chemicals that function as products, and materials on household chemicals subject to safety confirmation.
The following are special matters related to HCPs subject to safety confirmation subject to approval.
1) Products transferred from the Ministry of Food and Drug Safety will be managed as HCPs subject to safety confirmation during the grace period for approval of biocidal products and will be managed as biocidal products after the end of the grace period for approval of biocidal products.
2) Therefore, compliance with the regulations of biocides within the prescribed period must be observed at the same time as complying with the implementation of the ‘HCPs subject to approval’.
3) Compliance with Biocide regulation
- Notification of existing biocidal active substance
- Approval of Biocidal active substance
- Approval of biocidal products
- Indication standard of biocidal products
If you need more information, contact info@safety-as.com or +82 2 754 0600. Thank you.
▶ Join our group and get the recently news!: https://www.linkedin.com/groups/13911952/
#SafetyAssessmentSolution #Safety_As #K_BPR#householdchemicalproduct